Visby Sexual Health PCR Tests - Chlamydia/Gonorrhea/Trichomoniasis
Item Discontinued!
The Clinical Performance:
· CT - chlamydia: 97.4% PPA, 97.8 NPA
· NG - gonorrhea: 97.8% PPA, 99.1% NPA
· TV - trichomonas: 99.3% PPA, 96.7% NPA
Visby Coding Guide for STD Testing (edelberg.com)
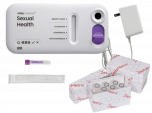
Visby Medical Sexual Health Test:
- CLIA waived PCR test for three STIs: Chlamydia, Gonorrhea, Trichomoniasis
- Delivers ~97% accuracy* in under 30 minutes
- Vaginal self-collected swab
- Improves antibiotic stewardship
The Visby Medical Sexual Health Test is a single-use (disposable), fully integrated, automated Polymerase Chain Reaction (PCR) in vitro diagnostic test intended for use in point-of-care or clinical laboratory settings for the rapid detection and differentiation of DNA from Chlamydia trachomatis, Neisseria gonorrhoeae, and Trichomonas vaginalis in self-collected female vaginal swab specimens using the Visby Medical Sexual Health Vaginal Specimen Collection Kit in a health care setting. The test results are to aid in the diagnosis of symptomatic or asymptomatic infections with Chlamydia trachomatis, Neisseria gonorrhoeae, and Trichomonas vaginalis.