Search results for: 'quidel quickvue chlamydia test'
-
 Quidel QuickVue Chlamydia TestsSKU :QDL-0B006(25 Tests) Item Discontinued! Please see below for alternative product: Visby Sexual Health PCR Tests - Chlamydia...
Quidel QuickVue Chlamydia TestsSKU :QDL-0B006(25 Tests) Item Discontinued! Please see below for alternative product: Visby Sexual Health PCR Tests - Chlamydia... -
 Quidel Chlamydia Swab PackSKU :QDL-20228(25 Swabs) 25 individually pouched sterile Rayon swabs. For use with QuickVue Chlamydia Test only....$16.00
Quidel Chlamydia Swab PackSKU :QDL-20228(25 Swabs) 25 individually pouched sterile Rayon swabs. For use with QuickVue Chlamydia Test only....$16.00 -
 Quidel QuickVue Influenza A/B TestsSKU :QDL-20183-IN(25 Tests) FREE Next Day Air Shipping on 4 or More Kits! QuickVue Influenza (FLU) A+B Test Kits (dipstick format), all...$359.00
Quidel QuickVue Influenza A/B TestsSKU :QDL-20183-IN(25 Tests) FREE Next Day Air Shipping on 4 or More Kits! QuickVue Influenza (FLU) A+B Test Kits (dipstick format), all...$359.00 -
 QuickVue Quidel iFOB Test KitSKU :QDL-20194(20 Tests) The QuickVue iFOB (immunochemical Fecal Occult Blood) test is an immunochemical device intended for the qual...$191.00
QuickVue Quidel iFOB Test KitSKU :QDL-20194(20 Tests) The QuickVue iFOB (immunochemical Fecal Occult Blood) test is an immunochemical device intended for the qual...$191.00 -
 Quidel QuickVue H. pylori gII (For Whole Blood)SKU :QDL-0W010(30 Tests) Quidel QuickVue H. pylori gII (For whole blood). The QuickVue H. pylori gII test is a lateral flow immuno ch...$350.00
Quidel QuickVue H. pylori gII (For Whole Blood)SKU :QDL-0W010(30 Tests) Quidel QuickVue H. pylori gII (For whole blood). The QuickVue H. pylori gII test is a lateral flow immuno ch...$350.00 -
 Quidel QuickVue H. pylori gII (For whole blood)SKU :QDL-0W009(10 Tests) Out of Stock: Lead time is 60-90 days. See suggested alternatives below: CLIA-05-9476 ALF-05-9475 Q...$125.00
Quidel QuickVue H. pylori gII (For whole blood)SKU :QDL-0W009(10 Tests) Out of Stock: Lead time is 60-90 days. See suggested alternatives below: CLIA-05-9476 ALF-05-9475 Q...$125.00 -
 Quidel QuickVue FLU A/B and RSV Promo BundleSKU :QDL-FLURSV-PROMOThis package bundle includes the following: 1 Box of Quidel QuickVue FLU A/B Tests (25 Tests) 1 Box of Quid...$599.00
Quidel QuickVue FLU A/B and RSV Promo BundleSKU :QDL-FLURSV-PROMOThis package bundle includes the following: 1 Box of Quidel QuickVue FLU A/B Tests (25 Tests) 1 Box of Quid...$599.00 -
 Quidel QuickVue Strep A TestSKU :QDL-20122(25 Tests) Item Out of Stock! Please see alternative item below: QuickVue In-Line Strep A (25/Box) (cliawaived.com) ...
Quidel QuickVue Strep A TestSKU :QDL-20122(25 Tests) Item Out of Stock! Please see alternative item below: QuickVue In-Line Strep A (25/Box) (cliawaived.com) ... -
 Quidel QuickVue RSV Test KitSKU :QDL-20193(20 Tests) In Stock - Ships Immediately! FREE Next Day Air Shipping on 4 or More Kits! Respiratory Syncytial Virus (R...$260.00
Quidel QuickVue RSV Test KitSKU :QDL-20193(20 Tests) In Stock - Ships Immediately! FREE Next Day Air Shipping on 4 or More Kits! Respiratory Syncytial Virus (R...$260.00 -
 Visby Sexual Health PCR Tests - Chlamydia/Gonorrhea/TrichomoniasisSKU :VSB-PS-400372(20 Tests & 1 Power Cord Adapter) The first true PCR point-of-care device for STI that’s all-in-one and requires ...$1,520.00
Visby Sexual Health PCR Tests - Chlamydia/Gonorrhea/TrichomoniasisSKU :VSB-PS-400372(20 Tests & 1 Power Cord Adapter) The first true PCR point-of-care device for STI that’s all-in-one and requires ...$1,520.00 -
 Quidel DipStick Strep A TestSKU :QDL-20108(50 Tests) The QuickVue Dipstick Strep A Test detects Group A Streptococcal antigen directly from patient throat swab...$89.95
Quidel DipStick Strep A TestSKU :QDL-20108(50 Tests) The QuickVue Dipstick Strep A Test detects Group A Streptococcal antigen directly from patient throat swab...$89.95 -
 QuickVue iFOB Test (50 Test/Kit) Acute Care PackSKU :QDL-20201This item is currently out of stock and lead time for new stock is 2-3 weeks. Orders are being accepted but lead tim...$465.00
QuickVue iFOB Test (50 Test/Kit) Acute Care PackSKU :QDL-20201This item is currently out of stock and lead time for new stock is 2-3 weeks. Orders are being accepted but lead tim...$465.00 -
 QuickVue At-Home Over-the-Counter COVID-19 TestsSKU :QDL-20402(2 Tests per Box) This item is currently out of stock and we recommend purchasing ACON Flowflex and/or OnGO Rapid Ho...
QuickVue At-Home Over-the-Counter COVID-19 TestsSKU :QDL-20402(2 Tests per Box) This item is currently out of stock and we recommend purchasing ACON Flowflex and/or OnGO Rapid Ho... -
 Quidel Triage Total 5 Calibration VerificationSKU :QDL-88755Calibration Verification Triage® Total 5 CK-MB, Myoglobin, Troponin I, BNP, D-Dimer 2 X 0.25 mL Each Level For Triage®...
Quidel Triage Total 5 Calibration VerificationSKU :QDL-88755Calibration Verification Triage® Total 5 CK-MB, Myoglobin, Troponin I, BNP, D-Dimer 2 X 0.25 mL Each Level For Triage®... -
 Quidel Triage Cardiac PanelSKU :QDL-97000HS(25 Tests) We price match! Please call for details.The Triage® Cardiac Panel is a rapid, point of care fluorescence im...$775.00
Quidel Triage Cardiac PanelSKU :QDL-97000HS(25 Tests) We price match! Please call for details.The Triage® Cardiac Panel is a rapid, point of care fluorescence im...$775.00 -
 QuickVue One-Step hCG-ComboSKU :QDL-20110(50 Per Kit) QuickVue One-Step hCG-Combo Pregnancy Testing Kits. The QuickVue One Step hCG Combo test is a sensitive im...$106.50
QuickVue One-Step hCG-ComboSKU :QDL-20110(50 Per Kit) QuickVue One-Step hCG-Combo Pregnancy Testing Kits. The QuickVue One Step hCG Combo test is a sensitive im...$106.50 -
 QuickVue One-Step hCG-UrineSKU :QDL-20109(25 Per Kit) QuickVue One-Step hCG-Urine Test. Rapid pregnancy testing kit, the QuickVue One Step hCG Urine test is a s...$54.00
QuickVue One-Step hCG-UrineSKU :QDL-20109(25 Per Kit) QuickVue One-Step hCG-Urine Test. Rapid pregnancy testing kit, the QuickVue One Step hCG Urine test is a s...$54.00 -
 QuickVue One-Step hCG-ComboSKU :QDL-179(90 Tests/Kit) QuickVue One-Step hCG-Combo Pregnancy Testing Kits. The QuickVue One Step hCG Combo test is a sensitive ...$225.00
QuickVue One-Step hCG-ComboSKU :QDL-179(90 Tests/Kit) QuickVue One-Step hCG-Combo Pregnancy Testing Kits. The QuickVue One Step hCG Combo test is a sensitive ...$225.00 -
 Quidel Triage Cardiac Panel, Troponin ISKU :QDL-97021HS(25 Test/Kit) The Quidel Triage Cardiac Panel is a rapid, point of care fluorescence immunoassay used with the Qu...$599.00
Quidel Triage Cardiac Panel, Troponin ISKU :QDL-97021HS(25 Test/Kit) The Quidel Triage Cardiac Panel is a rapid, point of care fluorescence immunoassay used with the Qu...$599.00 -
 Quidel Triage Total 5 Control 1SKU :QDL-88753(5 Vials/Box) Triage Kit, Control Level 1, TC5 includes BNP, D DIMER, CK-MB, MYO, TNI Item must be kept Frozen. Discl...
Quidel Triage Total 5 Control 1SKU :QDL-88753(5 Vials/Box) Triage Kit, Control Level 1, TC5 includes BNP, D DIMER, CK-MB, MYO, TNI Item must be kept Frozen. Discl... -
 Quidel Triage TOX Drug ScreenSKU :QDL-94600(25 Tests) The Quidel Triage TOX Drug Screen, 94600 is a fluorescence immunoassay to be used with the Quidel Tria...$825.00
Quidel Triage TOX Drug ScreenSKU :QDL-94600(25 Tests) The Quidel Triage TOX Drug Screen, 94600 is a fluorescence immunoassay to be used with the Quidel Tria...$825.00 -
 Quidel Triage Total 5 Control 2SKU :QDL-88754(5 Vials/Box) Triage Kit, Control Level 2, TC5 includes BNP, D DIMER, CK-MB, MYO, TNI Item must be kept Frozen. Discl...
Quidel Triage Total 5 Control 2SKU :QDL-88754(5 Vials/Box) Triage Kit, Control Level 2, TC5 includes BNP, D DIMER, CK-MB, MYO, TNI Item must be kept Frozen. Discl... -
 QuickVue SARS Antigen FIA DipstickSKU :QDL-20387(25 Tests per Kit) * Free Next Day Air when you buy 5 or more kits! Current stock Expires January 2025. No Returns or ...$175.00
QuickVue SARS Antigen FIA DipstickSKU :QDL-20387(25 Tests per Kit) * Free Next Day Air when you buy 5 or more kits! Current stock Expires January 2025. No Returns or ...$175.00 -
 QuickVue® Strep A Liquid ControlSKU :QDL-354(1 Positive, 1 Negative) Positive and negative liquid Strep A control set. For use with QuickVue Dipstick Strep A, Quic...$29.95
QuickVue® Strep A Liquid ControlSKU :QDL-354(1 Positive, 1 Negative) Positive and negative liquid Strep A control set. For use with QuickVue Dipstick Strep A, Quic...$29.95 -
 QuickVue hCG Control Set (Urine)SKU :QDL-272(Negative + Positive) The hCG Control Set – Urine is intended for use with QuickVue+ hCG Combo, QuickVue hCG Urine or...$32.95
QuickVue hCG Control Set (Urine)SKU :QDL-272(Negative + Positive) The hCG Control Set – Urine is intended for use with QuickVue+ hCG Combo, QuickVue hCG Urine or...$32.95 -
 QuickVue + Mono (20/Box)SKU :QDL-20121(20 Tests) Discontinued. See suggested alternatives below: LifeSign Status Mono Tests (cliawaived.com) OSOM Mono ...
QuickVue + Mono (20/Box)SKU :QDL-20121(20 Tests) Discontinued. See suggested alternatives below: LifeSign Status Mono Tests (cliawaived.com) OSOM Mono ... -
 Swab Pack (Dipstick & QuickVue+) 50/BagSKU :QDL-20227(50 Swabs) 50 individually pouched sterile Rayon swabs. For use with QuickVue Dipstick Strep A test and QuickVue+ Strep...$19.95
Swab Pack (Dipstick & QuickVue+) 50/BagSKU :QDL-20227(50 Swabs) 50 individually pouched sterile Rayon swabs. For use with QuickVue Dipstick Strep A test and QuickVue+ Strep...$19.95 -
 QuickVue Strep A Control SwabSKU :QDL-345(6 Positive, 6 Negative) Positive and negative control swab set. Includes (6) positive and (6) negative control swabs....$29.95
QuickVue Strep A Control SwabSKU :QDL-345(6 Positive, 6 Negative) Positive and negative control swab set. Includes (6) positive and (6) negative control swabs....$29.95 -
 QuickVue In-Line Strep A (25/Box)SKU :QDL-343(25 Tests per Box) The QuickVue In-Line Strep A Test uses a unique in-line extraction to detect group A streptococcal a...$89.95
QuickVue In-Line Strep A (25/Box)SKU :QDL-343(25 Tests per Box) The QuickVue In-Line Strep A Test uses a unique in-line extraction to detect group A streptococcal a...$89.95 -
 QuickVue One-Step hCG-ComboSKU :QDL-178(30/Box) QuickVue One-Step hCG-Combo Pregnancy Testing Kits. The QuickVue One Step hCG Combo test is a sensitive immuno...$79.00
QuickVue One-Step hCG-ComboSKU :QDL-178(30/Box) QuickVue One-Step hCG-Combo Pregnancy Testing Kits. The QuickVue One Step hCG Combo test is a sensitive immuno...$79.00 -
 Visby Sexual Health Tests - Collection KitSKU :VSB-PS-000715(50 Kits) For use with Visby Sexual Health PCR Tests - Chlamydia/Gonorrhea/Trichomoniasis (cliawaived.com). Sexual ...$126.00
Visby Sexual Health Tests - Collection KitSKU :VSB-PS-000715(50 Kits) For use with Visby Sexual Health PCR Tests - Chlamydia/Gonorrhea/Trichomoniasis (cliawaived.com). Sexual ...$126.00 -
 Visby Medical Sexual Health - Starter KitSKU :VSB-PS-400515Starter Kit Includes: Customer Commitment: 4...$1,800.00
Visby Medical Sexual Health - Starter KitSKU :VSB-PS-400515Starter Kit Includes: Customer Commitment: 4...$1,800.00 -
 QuikVue iFOB Test Control SetSKU :QDL-20197(1 Set) The QuickVue iFOB Control Set is intended for use with the QuickVue iFOB (immunochemical Fecal Occult Blood) te...$42.00
QuikVue iFOB Test Control SetSKU :QDL-20197(1 Set) The QuickVue iFOB Control Set is intended for use with the QuickVue iFOB (immunochemical Fecal Occult Blood) te...$42.00 -
 FREND TESTOSTERONE KitSKU :FREND-TEST-FRTE-020(20 Tests) The onsite TESTOSTERONE kit for use with FREND immunoassay system. Contains 20 test cartridges per box. ...$274.45
FREND TESTOSTERONE KitSKU :FREND-TEST-FRTE-020(20 Tests) The onsite TESTOSTERONE kit for use with FREND immunoassay system. Contains 20 test cartridges per box. ...$274.45 -
 Sofia SARS-Antigen Test KitSKU :QDL-20374(25 Tests per Kit) For use with Sofia 2 and Sofia LOWER PRICING MAY BE AVAILABLE TO THOSE CUSTOMERS THAT SIGN SOFIA ...$550.00
Sofia SARS-Antigen Test KitSKU :QDL-20374(25 Tests per Kit) For use with Sofia 2 and Sofia LOWER PRICING MAY BE AVAILABLE TO THOSE CUSTOMERS THAT SIGN SOFIA ...$550.00 -
 Flowflex COVID -19 Antigen Home Test (50 test pack)SKU :AC-L031-125P5-50(50 test pack) The Flowflex™ COVID-19 Antigen Home Test is all you need to determine your family’s Covid-19 status...$266.67
Flowflex COVID -19 Antigen Home Test (50 test pack)SKU :AC-L031-125P5-50(50 test pack) The Flowflex™ COVID-19 Antigen Home Test is all you need to determine your family’s Covid-19 status...$266.67 -
 Test Yourself @ Home - Home Pregnancy TestSKU :TY@H-HCG-M(1 Test) The Test Yourself @ Home (TY@H) Home Pregnancy Test is a self-performing immunoassay designed for the qualitati...$9.99
Test Yourself @ Home - Home Pregnancy TestSKU :TY@H-HCG-M(1 Test) The Test Yourself @ Home (TY@H) Home Pregnancy Test is a self-performing immunoassay designed for the qualitati...$9.99 -
 QuikVue iFOB Test Kit (100/Kit)SKU :QDL-20205(100 Tests) The QuickVue iFOB (immunochemical Fecal Occult Blood) Test is an immunochemical device intended for th...$610.00
QuikVue iFOB Test Kit (100/Kit)SKU :QDL-20205(100 Tests) The QuickVue iFOB (immunochemical Fecal Occult Blood) Test is an immunochemical device intended for th...$610.00 -
 Flowflex COVID -19 Antigen Home Test (25 test pack)SKU :AC-L031-125P5-25FlowFlex Home Covid test kit (25 test pack) ITEM DISCONTINUED We are sorry to inform you that this product has been di...
Flowflex COVID -19 Antigen Home Test (25 test pack)SKU :AC-L031-125P5-25FlowFlex Home Covid test kit (25 test pack) ITEM DISCONTINUED We are sorry to inform you that this product has been di... -
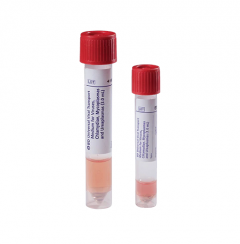 Universal Viral Transport Vials BD‚™ Plastic 3 mL Screw CapSKU :BD-220220(50 per Pack) BD UVT vial, 3 mL. The BD universal viral transport (UVT) system is designed to transport viruses, chla...
Universal Viral Transport Vials BD‚™ Plastic 3 mL Screw CapSKU :BD-220220(50 per Pack) BD UVT vial, 3 mL. The BD universal viral transport (UVT) system is designed to transport viruses, chla... -
 Universal Viral Transport Kit BD 3mL VialsSKU :BD-220531(50 Per Pack) BD UVT 3-mL collection kit with flexible minitip flocked swab....
Universal Viral Transport Kit BD 3mL VialsSKU :BD-220531(50 Per Pack) BD UVT 3-mL collection kit with flexible minitip flocked swab.... -
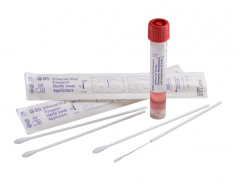
 Clearview Female Collection KitSKU :ALERE-135315(20 Per Kit) Discontinued, please click Request a Quote to inquire about a substitute item. Clearview Female Collectio...
Clearview Female Collection KitSKU :ALERE-135315(20 Per Kit) Discontinued, please click Request a Quote to inquire about a substitute item. Clearview Female Collectio... -
 Accula COVID-19 Test PromoSKU :MB-ACCULA-SARS-PROMOProduct Discontinued. See Below For Alternative Products: 3EO Health COVID-19 Molecular Test Starter Kit (cliawaive...
Accula COVID-19 Test PromoSKU :MB-ACCULA-SARS-PROMOProduct Discontinued. See Below For Alternative Products: 3EO Health COVID-19 Molecular Test Starter Kit (cliawaive... -
 NUVETEST™ Rapid Acidity Test Bacterial Vaginosis (BV) Test KitSKU :NUV-23710-008-25(25 tests) A one-step detection test for vaginitis associated with bacterial vaginosis (BV) and trichomonas....$79.00
NUVETEST™ Rapid Acidity Test Bacterial Vaginosis (BV) Test KitSKU :NUV-23710-008-25(25 tests) A one-step detection test for vaginitis associated with bacterial vaginosis (BV) and trichomonas....$79.00 -
 Solana Strep Complete Assay Rapid Test KitSKU :QDL-M305(48/Kit) Features The Solana Strep Complete Assay is a rapid in vitro diagnostic test for the qualitative detection ...
Solana Strep Complete Assay Rapid Test KitSKU :QDL-M305(48/Kit) Features The Solana Strep Complete Assay is a rapid in vitro diagnostic test for the qualitative detection ... -
 Solana GAS Assay Rapid Test Kit for Strep ASKU :QDL-M301(48/Kit) The Solana GAS Assay is a rapid in vitro diagnostic test for the qualitative detection of Group A B-hemolyti...$1,020.00
Solana GAS Assay Rapid Test Kit for Strep ASKU :QDL-M301(48/Kit) The Solana GAS Assay is a rapid in vitro diagnostic test for the qualitative detection of Group A B-hemolyti...$1,020.00 -
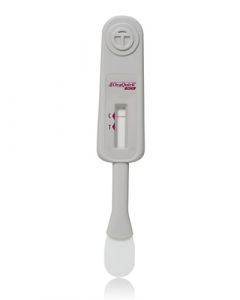 OraQuick HCV Rapid Antibody TestSKU :ORA-1001-0181(25 Tests) Now CLIA-waived! The OraQuick® HCV test is the FIRST FDA approved test for detecting HCV antibodies in fin...$437.50
OraQuick HCV Rapid Antibody TestSKU :ORA-1001-0181(25 Tests) Now CLIA-waived! The OraQuick® HCV test is the FIRST FDA approved test for detecting HCV antibodies in fin...$437.50


