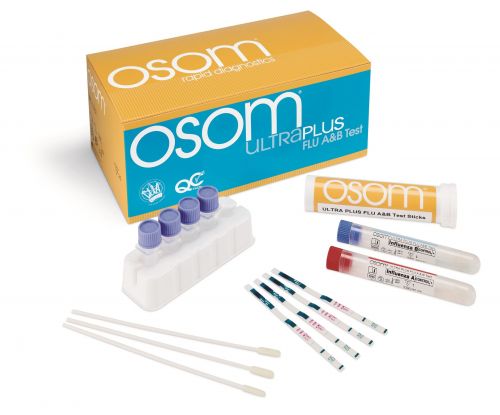
OSOM Ultra Plus Flu A&B Test Kit
SKU
GZM-1032
$289.95
(25 Tests)
Free Ground Shipping on 5 or More Boxes!
New Stock! - Long Expiration Date of February 2027!
The OSOM Ultra Plus Flu A & B Test is an in vitro rapid qualitative test that detects influenza type A and type B nucleoprotein antigens directly from nasal swab and nasopharyngeal swab specimens obtained from patients with signs and symptoms of respiratory infection.
- Meets the Influenza reclassification requirements for rapid antigen tests*
- Influenza A
- Sensitivity: 90.3%
- Specificity: 96.7%
- Influenza B
- Sensitivity: 88%
- Specificity: 99.2%
- Influenza A
*Refer to the Package Insert for additional performance claims.
- Simple CLIA-waived procedure with pre-measured extraction buffer for swab samples
- Accurate, differentiated interpretation of results in 10 minutes to test and treat patients in one office visit
Product Availability: USA
Sample Type: Nasal swab and nasopharyngeal swab
Storage and Stability:
- Patient samples should be processed in the Extraction Buffer vial as soon as possible or within 8 hours after collection.
- Patient swab may be stored at room temperature (15-30°C/59-86°F) for up to 8 hours, or refrigerated (2-8°C/36-46°F) for up to 24 hours prior to testing.
- Extracted sample (stored at room temperature) must be used within 2 hours.
- Acceptable transport media:
BD Universal Viral Transport Medium
Remel Micro Test M4, M4RT, M5 M6 Medium
Bartels FlexTrans Medium
Refrigerated samples should come to room temperature before testing.