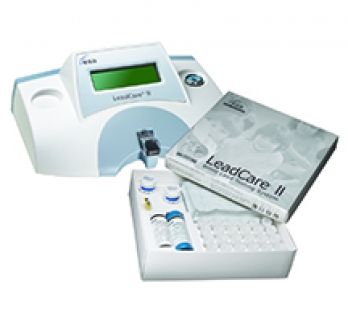
LeadCare II Analyzer Kit - includes 1 box of test ($399 value savings)
(1 System + 48 Tests)
Promotion: Get 1 box of 48 tests free with purchase of meter! Offer valid for sales in USA only!
The LeadCare II analyzer is a CLIA Waived system for the determination of lead in blood. The analyzer kit consists of the portable blood-lead testing instrument, AC power cord, AA batteries, User's Guide (English and Spanish), Quick reference guide, instructional video, and CDC blood collection video.
THIS ITEM IS NON REFUNDABLE AND NON RETURNABLE (please allow 5-10 business days for this item to ship from 3rd party supplier). Orders submitted for this product online cannot be cancelled, returned or refunded unless deemed defective or faulty by our supplier.
CLIA Waived LeadCare® II point-of-care system
The world's only CLIA-waived blood-lead system, LeadCare II improves clinical outcomes and is ideal for the physicians' offices, health clinics, or outreach-screening program.
Safe, fast, easy, and inexpensive to use, ESA developed LeadCare II with a grant from the U.S. Centers for Disease Control. No more waiting days for expensive lab analysis, or worrying about losing track of a child that needs follow-up care. LeadCare II delivers quantitative blood lead results equivalent to those reported by outside laboratories in just three minutes. It improves patient care by providing on-the-spot results, cuts your staffs' result-tracking and administrative time, and qualifies for reimbursement as a quantitative blood lead test (CPT code: 83655).
- Point-of-care results in just three minutes, eliminates multiple visits
- Simple to operate, no special training required
- Capillary samples (two drops, 50 ìl), easier for patient, parent, and staff
- Accuracy and sensitivity you can count, comparable to reference-lab tests
- Reimbursable, CPT code: 83655; per-test cost typically less than a reference-lab test
LeadCare II features:
- Small-9.0 in x 6.5 in x 3.5 in
- Lightweight, 2.4 lbs
- Portable, runs on four AA battery or AC adapter
- One-touch electronic calibration
- All reagents and supplies provided in a convenient test kit, stable at room temperature
- Completely safe, unique gold electrode sensor contains no mercury or other toxic materials
Qty: 1 Analyzer
CLIA: FDA Cleared / CLIA-Waived
Contact: (888)-882-7739
CPT Code*: 83655QW - Assay of lead
National Average Reimbursement 2019: $13.45
*All CPT codes are supplied for information purposes only and represent no statement; promise or guarantee by CLIAwaived Inc. that these codes will be appropriate or that reimbursement will be made. It is the responsibility of the service provider to confirm the appropriate coding required by their local Medicare carriers, fiscal intermediaries and commercial payors.